Schrodinger equation for hydrogen
Here we shall take the system to mean just the electrons.
#SCHRODINGER EQUATION FOR HYDROGEN UPDATE#
Since the proton is much more massive than the electron, we will assume throughout this chapter that the reduced mass equals the electron mass and the proton is located at the center of mass. The Schrodinger Equation for Hydrogen and Multi-Electron Systems Last update 17/1/10 The time-independent Schrodinger equation is always simply, HE (1) where E is the total energy of the system.

The length of \(r\) is the distance between the proton and the electron, and the direction of \(r\) and the direction of \(r\) is given by the orientation of the vector pointing from the proton to the electron. Using the Schrödinger equation tells you just about all you need to know about the hydrogen atom, and it’s all based on a single assumption: that the wave function must go to zero as r goes to infinity, which is what makes solving the Schrödinger equation possible. Hydrogen Atom Solution to the Schrodinger Equation Finally, the hydrogen atom solution has obvious applications to real-world physics, but in practice the situation for an electron around the nucleus of a hydrogen atom can be seen as pretty similar to the potential well problems. This reduced particle is located at \(r\), where \(r\) is the vector specifying the position of the electron relative to the position of the proton. The free-complement (FC) theory for solving the Schrdinger equation (SE) was applied to calculate the potential energy curves of the ground and excited. Import weave def Numerovc ( f, x0_, dx, dh_ ): code_Numerov = """ double h2 = dh*dh double h12 = h2/12. The Schrdinger equation of hydrogen molecules was solved essentially exactly and systematically for calculating the potential energy curves of the.


#SCHRODINGER EQUATION FOR HYDROGEN PLUS#
Our equation is minus h squared over 2m, d second dr squared-the radial equation-plus h squared l times l plus 1 over 2mr squared minus ze squared over r, u is equal to Eu. Schrodinger’s hydrogen atom The time-independent Schrodinger equation in three-dimensions may be written as The hydrogen atom may be represented by the Schrodinger equation with this centrosymmetric Coulomb potential.
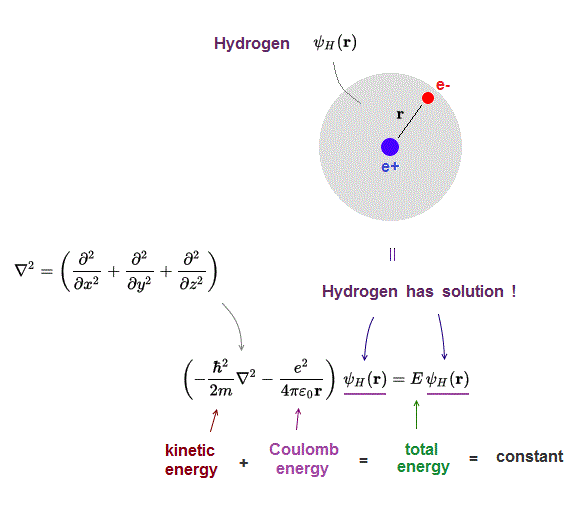
The implementation will follow these steps When dealing with a system of more than one particle, as with the hydrogen atom, center of mass coordinates are used to represent the system. So it's really unbelievable how much you can learn with the fine spectrum of the hydrogen atom.
